|
|
Post by jeff on May 15, 2016 22:48:56 GMT
Given renewable energy as a source for charging, the VRFB has the least damaging environmental impact. The electrolyte is the storage media, with the pumps and cells being the only 'disposable' parts of the system. For all intents and purposes, the electrolyte is endlessly recyclable. Being able to produce them would solve the issue of obtaining them, since household-size VRFBs exist, but there is no intent to sell/lease them to individuals, at least from the companies I have contacted so far. LCA of in-house producED SMAll-SIZED vanadium redox-flow batterY Authors Michele Dassisti www.academia.edu/24928016/LCA_of_in-house_producED_SMAll-SIZED_vanadium_redox-flow_batterY |
|
|
|
Post by jeff on May 15, 2016 22:56:57 GMT
|
|
|
|
Post by jeff on May 16, 2016 2:52:26 GMT
|
|
|
|
Post by jeff on May 16, 2016 3:12:08 GMT
|
|
|
|
Post by jeff on May 16, 2016 4:15:11 GMT
From the rustbelt: An iron-based flow battery phys.org/news/2011-05-rustbelt-iron-based-battery.htmlResearchers at Case Western Reserve University are mixing cheap and plentiful iron in benign solutions to create a flow battery – essentially an unwrapped battery that can be scaled up to hold and supply electricity to a home or an entire community. The goal is to produce a cheap and efficient system capable of storing energy from wind turbines and solar panels and supplying energy when wind wanes and the sun sets. The battery could also be integrated into a smart grid, charging up when usage is low then adding electricity when need is high. Read more at: phys.org/news/2011-05-rustbelt-iron-based-battery.html#jCp |
|
|
|
Post by jeff on May 16, 2016 4:18:20 GMT
Membranes for Redox Flow Battery Applications Abstract: The need for large scale energy storage has become a priority to integrate renewable energy sources into the electricity grid. Redox flow batteries are considered the best option to store electricity from medium to large scale applications. However, the current high cost of redox flow batteries impedes the wide spread adoption of this technology. The membrane is a critical component of redox flow batteries as it determines the performance as well as the economic viability of the batteries. The membrane acts as a separator to prevent cross-mixing of the positive and negative electrolytes, while still allowing the transport of ions to complete the circuit during the passage of current. An ideal membrane should have high ionic conductivity, low water intake and excellent chemical and thermal stability as well as good ionic exchange capacity. Developing a low cost, chemically stable membrane for redox flow cell batteries has been a major focus for many groups around the world in recent years. This paper reviews the research work on membranes for redox flow batteries, in particular for the all-vanadium redox flow battery which has received the most attention. www.mdpi.com/2077-0375/2/2/275/pdf |
|
|
|
Post by jeff on May 16, 2016 4:24:35 GMT
New Simpler Flow Battery for Grid Storage Developed at Stanford University One promising grid storage solution is redox flow battery technology, which uses two tanks of liquids functioning as anode and cathode. Pumps circulate the liquids to a reaction chamber where they are kept separate by a membrane that allows ions to pass back and forth while cycling the battery. It’s easily scalable, but very expensive to install and maintain. Stanford University and the Department of Energy have developed a flow battery that could prove to be an even better grid storage solution. The new lithium-polysulfide flow battery only uses an anode liquid with one pump and no membrane. The liquid passes by a lithium cathode to transfer ions during battery cycling. With half the parts, less expensive anode liquid, and no expensive membrane to maintain, the new battery can be scaled even easier than traditional redox flow batteries. 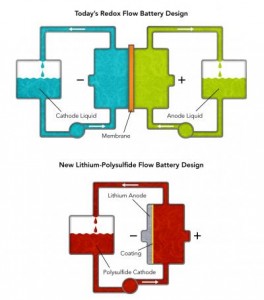 www.greenoptimistic.com/flow-battery-grid-storage-stanford-20130425/#.VzlKIzWsjGs www.greenoptimistic.com/flow-battery-grid-storage-stanford-20130425/#.VzlKIzWsjGs |
|
|
|
Post by jeff on May 16, 2016 5:10:34 GMT
A metal-fee organic-inorganic aqueous flow battery The QBFB comprised a mixture of commercially available and custom-made components. Pretreated 2 cm2, stacked (36) Toray carbon paper electrodes (each of which is about 7.5 mmuncompressed) were used on both sides of the cell. Nafion 212 (50 mmthick) was used as a proton-exchange membrane, and PTFE gasketing was used to seal the cell assembly. On the positive side of the cell, 120 ml of 3M HBrand 0.5MBr2 were used as the electrolyte solution in the fully discharged state; on the negative side, 1M2,7-AQDS in 1MH2SO4 was used. AQDS disodium salt was flushed twice through a column containingAmberlyst 15Hion-exchange resin to remove the sodium ions. Half-cell measurements were conducted using a Ag/ AgCl aqueous reference electrode (3M KCl filling solution), a Pt wire counter electrode and a 3-mm-diameter glassy carbon disk electrode. For theoretical calculations, the total free energies of molecules were obtained from first-principles quantum chemical calculations within density functional theory at the level of the generalized gradient approximation (GGA) using the PBE functional. Threedimensional conformer structures for each quinone/hydroquinonemoleculewere generatedusing the ChemAxon suite with up to 25 generated conformers permolecule using the Dreiding force field. Generated conformers were used as input structures for the density functional theory geometry optimization employed for determining the formation energy, which in turn is used to evaluate the reduction potential. In the QBFB cost calculation, a price of $4.74 per kilogram (eBioChem) was used for anthraquinone. To get the sulphonated form actually used here, anthraquinone must be reacted with oleum(H2SO4/SO3), which adds a negligible cost at scale; this cost is not included here. The price of bromine was $1.76 per kilogram, based on estimates from the US Geological Survey17. The cell voltage used to calculate costs here was 0.858 V. www.researchgate.net/profile/Alan_Aspuru-Guzik/publication/259630344_A_metal-fee_organic-inorganic_aqueous_flow_battery/links/0046352e9190a5f461000000.pdf?origin=publication_detail&ev=pub_int_prw_xdl&msrp=aLHzW4_nc_HCNmw8ctrnSROHsUGd5qP6kMHn8RGsx4Ls8VHVJKdKHpyYLXlhc44ldRoGN0nTrNLqLt2C5arJx2TxLdRv1GAnr6CZTqIxr4E.S0qdApbEB2gXcZFYsYY1R1j8g2kZxOJhFqZInXvrhecZEJWAw_kK09Rh1-BmgbvxEvGox5se0lIuNquM8bK5Ww.qy3GZMMVb2iEh_OSw70mhmbeEAiV3WKIdzyEnEL29ge-BLxSUVd6xwr2Rn0_Q766cJbX1qciR439za0RK8UOew |
|
|
|
Post by jeff on May 16, 2016 5:17:54 GMT
Making a vanadium redox flow battery
|
|
|
|
Post by jeff on May 16, 2016 6:10:16 GMT
High–energy density nonaqueous all redox flow lithium battery enabled with a polymeric membrane Abstract Redox flow batteries (RFBs) are considered one of the most promising large-scale energy storage technologies. However, conventional RFBs suffer from low energy density due to the low solubility of the active materials in electrolyte. On the basis of the redox targeting reactions of battery materials, the redox flow lithium battery (RFLB) demonstrated in this report presents a disruptive approach to drastically enhancing the energy density of flow batteries. With LiFePO4 and TiO2 as the cathodic and anodic Li storage materials, respectively, the tank energy density of RFLB could reach ~500 watt-hours per liter (50% porosity), which is 10 times higher than that of a vanadium redox flow battery. The cell exhibits good electrochemical performance under a prolonged cycling test. Our prototype RFLB full cell paves the way toward the development of a new generation of flow batteries for large-scale energy storage.  www.researchgate.net/publication/284912572_High-energy_density_nonaqueous_all_redox_flow_lithium_battery_enabled_with_a_polymeric_membranewww.sciencemag.org/news/2015/11/new-type-flow-battery-can-store-10-times-energy-next-best-device www.researchgate.net/publication/284912572_High-energy_density_nonaqueous_all_redox_flow_lithium_battery_enabled_with_a_polymeric_membranewww.sciencemag.org/news/2015/11/new-type-flow-battery-can-store-10-times-energy-next-best-devicetechxplore.com/pdf368092584.pdf There is still one major hurdle to overcome, as the team acknowledges, the RFLB is far too slow to be useful in a commercial application, 10,000 times slower than conventional RFBs—the group believes they will be able to speed it up though, by modifying the membrane and charge carrying mediators. |
|
|
|
Post by jeff on May 16, 2016 6:28:01 GMT
|
|
|
|
Post by jeff on May 16, 2016 6:38:08 GMT
All-Iron Redox Flow Battery Tailored for Off-Grid Portable Applications An all-iron redox flow battery is proposed and developed for end users without access to an electricity grid. The concept is a low-cost battery which the user assembles, discharges, and then disposes of the active materials. The design goals are: (1) minimize upfront cost, (2) maximize discharge energy, and (3) utilize non-toxic and environmentally benign materials. These are different goals than typically considered for electrochemical battery technology, which provides the opportunity for a novel solution. The selected materials are: low-carbon-steel negative electrode, paper separator, porous-carbon-paper positive electrode, and electrolyte solution containing 0.5 M Fe2 (SO4 )3 active material and 1.2 M NaCl supporting electrolyte. With these materials, an average power density around 20 mW cm(-2) and a maximum energy density of 11.5 Wh L(-1) are achieved. A simple cost model indicates the consumable materials cost US$6.45 per kWh(-1) , or only US$0.034 per mobile phone charge. www.researchgate.net/publication/284279832_All-Iron_Redox_Flow_Battery_Tailored_for_Off-Grid_Portable_Applications(Full paper requested) |
|
|
|
Post by jeff on May 16, 2016 21:50:15 GMT
A zinc-iron redox-flow battery under $100/kWh of system capital cost Abstract Redox flow batteries (RFBs) are one of the most promising scalable electricity-storage systems to address the intermittency issues of renewable energy sources such as wind and solar. The prerequisite for RFBs to be economically viable and widely employed is their low cost. Here we present a new zinc-iron (Zn-Fe) RFB based on double-membrane triple-electrolyte design that is estimated to have under $100 per kW h system capital cost. Such a low cost is achieved by a combination of inexpensive redox materials (i.e., zinc and iron) and high cell performance (e.g., 676 mW cm-2 power density). Engineering of the cell structure is found to be critical to enable the high power density. Our cost model shows that a Zn-Fe RFB demonstrates the lowest cost among some notable RFBs and could reach the 2023 cost target set by the U.S. Department of Energy ($150 per kW h).  www.researchgate.net/publication/282522115_A_zinc-iron_redox-flow_battery_under_100kWh_of_system_capital_cost www.researchgate.net/publication/282522115_A_zinc-iron_redox-flow_battery_under_100kWh_of_system_capital_cost |
|
|
|
Post by jeff on May 17, 2016 4:59:44 GMT
While that all sounds fine and dandy, the upfront cost for even a 5 day reserve is as much as some automobiles.
Hawaii ave is just over 6,000 kWh/year/household
Round up and say 16.5 kWh/day, some 80+kWh capacity, at $250/kWh ~ $20k
|
|